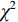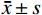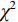The value of constructing a nomogram model based on arterial-phase imaging radiomics features combined with clinical-CT features in differentiating lung squamous carcinoma and adenocarcinoma
-
摘要:
目的:探讨基于动脉期影像组学特征联合临床-CT特征构建列线图模型在鉴别肺鳞癌(SCC)与肺腺癌(ADC)中的应用价值。方法:回顾性收集2021年8月-2023年9月在我院进行穿刺病理活检或手术的肺癌患者85例作为训练集,并同时收集2023年5月-2024年6月我院经病理证实的肺癌患者40例作为验证集,所有患者均行胸部CT增强检查。根据病理结果将训练集分为SCC组(n=29)和ADC组(n=56)。比较两组患者一般临床资料和CT图像特征的差异,采用单因素和多因素Logistic回归分析筛选出独立预测因素,并构建临床-CT模型。应用ITK Snap软件提取训练集动脉期图像影像组学特征,依次采用组内相关系数(ICC)、Roruta特征筛选和最小绝对收缩和选择算子(LASSO)对提取的影像组学特征进行降维处理,筛选出有意义的特征,采用Logistic回归构建动脉期影像组学模型,并计算该模型的影像组学评分(Rad-score)。以多因素Logistic回归分析筛选出临床-CT特征有意义的自变量与Rad-score构建联合模型,并绘制列线图。应用ROC曲线、校正曲线、H-L检验、Delong检验及临床决策曲线(DCA)对临床-CT模型、影像组学模型及列线图模型进行评价。结果:单因素分析结果显示,分叶征、坏死空洞征均多于ADC组(均P<0.05),癌胚抗原(CEA)、血管集束征、胸膜牵拉及毛刺征均少于ADC组(均P<0.05)。将上述自变量纳入多因素Logistic进一步筛选,结果显示,CEA、分叶征、胸膜牵拉及毛刺征为独立危险因素,基于此构建临床-CT模型的训练集和验证集AUC值分别为0.623和0.786。影像组学特征经降维后共筛选出的有意义特征有8个,分别为一阶特征3个、二阶特征5个。经ROC曲线分析显示,影像组学模型训练集和验证集AUC值分别为0.830和0.846;列线图模型训练集和验证集AUC值分别为0.913和0.922。经Delong检验显示,列线图模型AUC值均明显高于临床-CT模型和影像组学模型(均P<0.05);Hosmer-Lemeshow检验结果显示,临床-CT模型、影像组学模型及列线图模型的拟合度均良好;校准曲线分析显示,列线图模型的预测概率曲线与理想曲线最接近,预测精准度更好;DCA分析结果显示,列线图模型的曲线下面积最大,临床净收益最高。结论:基于动脉期影像组学特征联合临床-CT特征构建列线图模型在鉴别SCC与ADC中具有一定的诊断价值,为无创鉴别SCC与ADC提供一种新的诊断方式。
Abstract:Objective: To investigate the value of constructing a nomogram model based on arterial-phase imaging radiomics features combined with clinical-CT features for the differentiation between squamous lung cancer (SCC) and adenocarcinoma of the lung (ADC). Methods: Retrospectively, 85 patients with lung cancer who underwent puncture pathology biopsy or surgery in our hospital, from August 2021 to September 2023, were collected as a training set. Concurrently, 40 patients with pathologically confirmed lung cancer in our hospital, from May 2023 to June 2024, were collected as a validation set. All patients underwent chest CT enhancement. The training set was divided into the SCC group (n=29) and the ADC group (n=56) based on the pathology. General clinical data and CT image characteristics of the two groups of patients were compared and differences were identified. Independent predictors were screened using unifactorial and multifactorial logistic regression analyses, and a clinical-CT model was constructed. ITK Snap software was applied to extract the radiomics features of the arterial-phase images, and the intragroup correlation coefficient (ICC), Roruta feature screening, and least absolute shrinkage and selection operator (LASSO) were sequentially used to downsize the extracted radiomics features, screen out the meaningful features, construct the arterial-phase image radiomics model using Logistic regression, and compute the model's image radiomics score (Rad-score). A multifactor logistic regression analysis was used to screen the independent variables with meaningful clinical-CT characteristics and Rad-score to construct a joint model, and a nomogram graph was plotted. ROC curves, calibration curves, H-L test, Delong test, and clinical decision curves (DCA) were applied to evaluate the clinical-CT, radiomics, and nomogram models. Results: The results of univariate analysis showed that there were more lobular signs and necrotic cavity signs, and fewer carcinoembryonic antigen (CEA), vascular cluster signs, pleural pulling, and burr signs in the SCC than in the ADC group (all P < 0.05). The above independent variables were included in the multifactorial Logistic analysis for further screening, and the results showed that CEA, lobular sign, pleural pull, and burr sign were independent risk factors. The area under the curve (AUC) values for the training and validation sets of the clinical-CT model constructed based on this were 0.623 and 0.786, respectively. A total of eight meaningful features were screened after dimensionality reduction of the radiomics features, which were three first-order features and five second-order features. The ROC curve analysis showed that the AUC values for the training and validation sets of the radiomics model were 0.830 and 0.846, respectively; and the AUC values for the training and validation sets of the nomogram model were 0.913 and 0.922, respectively. The Delong test showed that the AUC values of the nomogram model were all significantly higher than those of the clinical-CT model and the radiomics model (all P < 0.05); the Hosmer-Lemeshow test showed that the clinical-CT model, the radiomics model, and the nomogram model were all well fitted; calibration curve analysis showed that the predictive probability curve of the nomogram model was closest to the ideal curve, with better predictive accuracy; and DCA analysis showed that the AUC of the nomogram model was the largest, with the highest net clinical benefit. Conclusion: Constructing a nomogram model based on arterial-phase imaging radiomics features combined with clinical-CT features has some diagnostic value in differentiating SCC from ADC, providing a new diagnostic modality for noninvasive differentiation.
-
Keywords:
- Arterial phase /
- Radiomics /
- Clinical-CT features /
- Squamous cell carcinoma /
- Adenocarcinoma /
- Nomogram model
-
肺癌是我国发病率和死亡率均较高的恶性肿瘤之一,主要以非小细胞肺癌为主,约占80%以上,包括肺鳞癌(Squamous cell carcinoma,SCC)和肺腺癌(Adenocarcinoma,ADC)[1]。以往研究表明[2],SCC与ADC生物学行为各不相同,ADC恶性程度相对较高,更易发生转移,ADC主要以免疫靶向药物或放化疗联合治疗为主,而SCC主要以手术联合放疗为主,患者预后效果与治疗方式密切相关,因此治疗前明确患者肺癌病理类型极其重要。吕祥瑞[3]等研究发现,血清肿瘤标志物水平与肺癌病理类型具有确定的相关性,其对准确鉴别肺癌病理类型具有参考价值。张盼[4]等在SCC与ADC采用动态增强CT扫描下的影像特征与其病理类型的关系中研究表明,ADC多为均匀强化,而SCC多为不均匀强化,但缺乏定量参数的支持,其诊断效能不高。近年来,随着影像组学的不断发展,对肿瘤的影像学评价已从定性形态学特征转化为定量组学特征,该技术能够高通量提取肿瘤影像组学特征定量信息,准确有效地对肿瘤病理类型进行鉴别诊断[5~6],但是大部分学者都是采用单一影像组学模型进行相关研究。基于此,本研究通过动脉期影像组学特征联合临床-CT特征构建 Logistic 回归模型,并绘制列线图,旨在为进一步提高鉴别ADC与SCC的准确性提供新的思路。
1. 资料与方法
1.1 一般资料
选取我院2021年8月-2023年9月85例经手术或穿刺病理证实的肺癌患者作为训练集,其中,男52例,女33例,年龄51~86岁;SCC 29例、ADC 56例。同时收集2023年5月-2024年6月我院经病理证实的肺癌患者40例作为验证集,其中,男28例,女12例,年龄44~90岁,SCC 15例、ADC 25例。纳入标准:(1)所有患者病理诊断结果为SCC或ADC;(2)治疗前所有患者均进行胸部CT增强检查;(3)胸部无既往肿瘤史;排除标准:(1)甲亢、哮喘者;(2)严重肝肾功能不全及碘对比剂过敏者;(3)临床及影像资料收集不全者。
1.2 仪器与方法
1.2.1 胸部CT增强检查
采用美国GE公司256排CT扫描仪。所有患者均行胸部 CT增强扫描,主要扫描参数:管电压80~140 Kvp,管电流采用自动管电流调制技术(150~300 mA),扫描层厚及间距 5 mm,螺距0.987,转速0.8 s/r,矩阵512×512,准直器宽度8 cm,重建层厚及层间距采用0.625 mm。采用德国欧立奇双筒高压注射器,经肘正中静脉置入20 G套管针,注射流速2.5~3.0 mL/s,对比剂用量1.0~1.5 mL/kg。CT增强动脉期扫描延迟时间25 s,静脉期扫描延迟时间50 s。患者采取仰卧位头先进,双手举过头顶,自肺尖至扫描至肺底,扫描时嘱咐患者吸气屏气。
1.2.2 肿瘤血清指标检测
所有患者于入院第1天早晨空腹采集10 ml外周静脉血,采用转速
3000 r/min离心5 min后分离上清,应用罗氏公司生产的全自动电化学发光仪及配套试剂盒对血清进行检测,测定神经元特异性烯醇化酶(neuron specific enolase,NSE)、癌胚抗原(carcinoembryonic antigen,CEA)、糖类抗原199(carbohydrate antigen199,CA199)、糖抗原242(carbohydrate antigen,CA242)。1.3 图像分析
将CT影像数据传至GEAW4.7工作站,由2名高级职称以上副主任医师采用双盲法在病灶最大层面分别采用肺窗(窗宽:
1200 Hu,窗位-600 Hu)和纵膈窗(窗宽:450 Hu,窗位:50 Hu)进行CT影像特征分析,意见不一致时,通过协商达成一致。CT影像特征标准[7-8]:(1)血管集束征:病灶周围可见一支或多支血管结构受病灶的牵拉向病灶方向集中或通过病灶或在病灶的边缘截断的表现;(2)坏死空洞:病灶内液化坏死和气体形成;(3)毛刺征:病灶边缘出现数目不等,呈放射粗细不均,呈条状排列的线状影;(4)分叶征:病灶的弦弧距和弦长之比值>0.4;(5)空泡征:病灶内部见直径<5 mm的细小圆形或卵圆形气体密度影;(6)胸膜牵拉征:病灶与脏层胸膜之间出现长条状密度增高影并有牵拉胸膜的表现。1.4 影像组学分析
1.4.1 病灶分割及特征提取
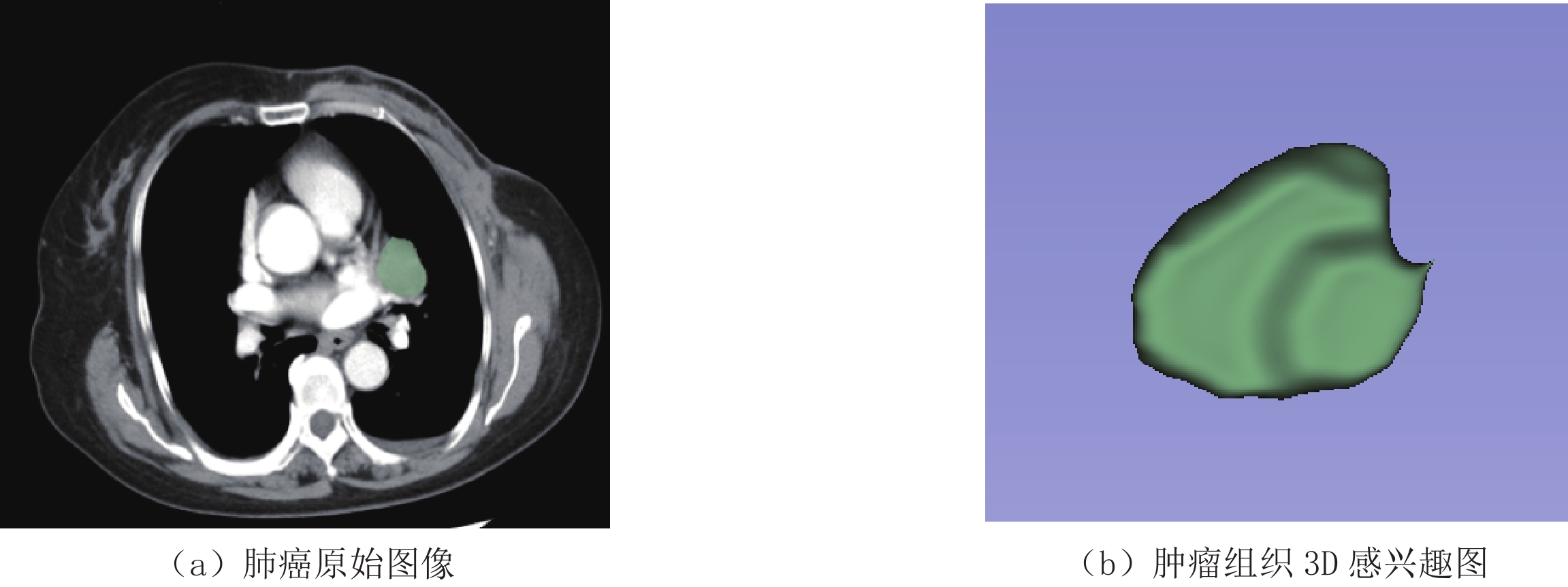
由于动脉期影像对评估肿瘤血供和强化程度更敏感,能够对肿瘤进行定性和定量分析,帮助医师鉴别肿瘤良恶性、分型及评估病情进展等;而静脉期影像通常用于评估肿瘤周围淋巴结是否转移和肺静脉血流等方面,故本研究只提取了肿瘤动脉期影像组学特征。将肿瘤动脉期图像以Dicom格式导入ITK-Snap软件,由2名高级职称以上放射诊断医师对肿瘤进行分割,采用手动方式对肿瘤病灶最大层面进行勾画,尽可能覆盖所有肿瘤组织,包括肿瘤内部囊变坏死区等,得到3D感兴趣区图(Volumes of interest,VOI)(图1)。随后在ITK-Snap平台上对分割的肿瘤图像进行影像组学特征提取,共提取动脉期影像组学特征786个。
1.4.2 特征降维及筛选
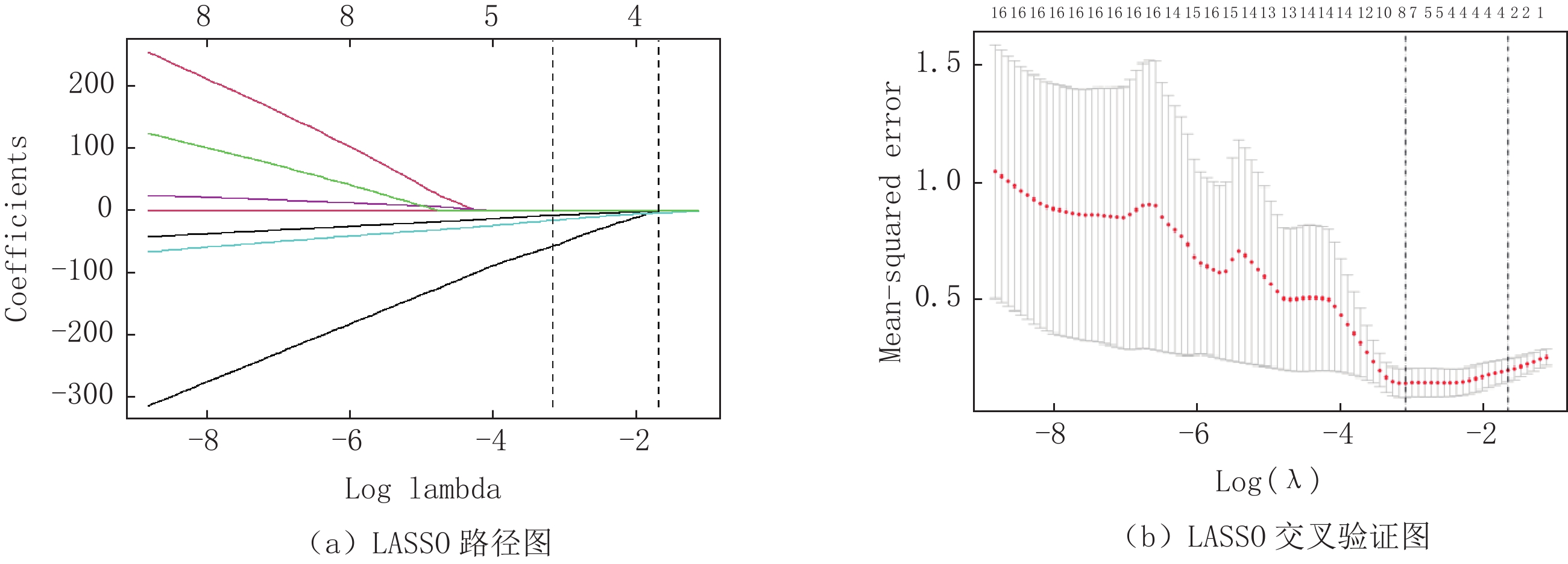
采用组内相关系数(intraclass correlation coemcient,ICC)对提取的影像组学特征一致性进行评价,保留ICC≥0.80的影像组学特征,共保留253个,然后采用Boruta特征筛选方法进行300次迭代,对253个组学特征进行二次降维,保留了59个特征,最后采用最小绝对收缩与选择算子(Least absolute shrinkage and selection operator,LASSO)通过十倍交叉验证得到最佳影像组学特征(图2),并建立影像组学评分(Radiomics score,Rad-score)。
1.5 统计学分析
应用R语言和SPSS 21.0软件对数据分析,服从正态分布计量资料以(
$\bar x\pm s $ )表示,行t检验,不服从正态分布计量资料以中位数,四分位数[M,(P25,P75)]表示,采用Mann-Whitney U检验;计数资料以n(%)表示,行$\chi^2 $ 检验。将有意义的临床-CT指标与最佳影像组学特征纳入Logistic回归构建联合模型,并绘制列线图。应用受试者操作特征(receiver operating characteristic,ROC)曲线、校正曲线和临床决策曲线(decision curve analysis,DCA)评价各模型的诊断效能,并采用Delong检验比较各模型曲线下面积(area under curve,AUC)差异;最后,应用Hosmer-Lemeshow检验评价各模型的拟合优度,以P<0.05为差异有统计学意义。2. 结果
2.1 训练集两组患者临床资料和CT影像特征结果比较
两组患者性别、年龄、吸烟史、家族肿瘤史、空泡征、肿瘤位置、NSE、CA242及CA199差异均无统计学意义(均P>0.05);SCC组分叶征、坏死空洞征均多于ADC组(均P<0.05),癌胚抗原(CEA)、血管集束征、胸膜牵拉及毛刺征少于ADC组(均P<0.05),见表1。
表 1 训练集两组患者临床资料与CT影像特征结果比较($\bar x\pm s $ )[n(%)]Table 1. Comparison of clinical data and results of CT imaging characteristics between the two groups of patients in the training set指标 SCC组(n=29) ADC组(n=56) t/x2/Z P 年龄 72.21±9.27 67.84±9.64 1.206 0.144 性别 1.231 0.121 男 24(82.76) 28(50.00) 女 5(17.24) 28(50.00) 吸烟史 0.188 0.665 是 19(65.52) 34(60.71) 否 10(34.48) 22(39.29) 家族肿瘤史 0.197 0.657 是 12(41.38) 26(46.43) 否 17(58.62) 30(53.57) NSE(ng/mL) 4.56(3.04, 7.38) 6.24 (4.12, 8.33) 0.413 0.115 CEA(ng/ml) 96.53±8.15 198.22±21.51 6.495 <0.001 CA242(IU/ml) 28.42±4.37 37.49±5.84 1.037 0.189 CA199(U/mL) 51.17±9.53 65.27±10.51 0.202 0.565 坏死空洞 有 25(86.21) 13(23.21) 35.325 <0.001 无 2(6.89) 43(76.79) 毛刺征 有 10(34.48) 45(80.36) 17.606 <0.001 无 19(65.52) 11(19.64) 分叶征 有 26(89.66) 10(17.86) 40.340 <0.001 无 3(10.34) 46(82.14) 空泡征 有 22(75.86) 32(57.14) 2.890 0.089 无 7(24.14) 24(42.86) 胸膜牵拉 有 16(55.17) 44(78.57) 4.574 0.032 无 13(44.83) 12(21.43) 血管集束征 有 13(44.83) 41(73.21) 6.645 0.010 无 16(55.17) 15(26.79) 肿瘤位置 上叶 18(62.07) 30(53.57) 2.011 0.093 中叶 3(10.34) 9(16.07) 下叶 8(27.59) 17(30.36) 2.2 临床-CT模型构建
在训练集中,以单因素分析具有统计学差异的指标作为自变量进行二元多因素Logistic回归分析,结果显示,CEA、分叶征、胸膜牵拉及毛刺征是鉴别ADC与SCC的独立影响因素,其回归方程为 P =-5.977+0.005×CEA+1.992×分叶征+1.413×胸膜牵拉+1.466×毛刺征,见表2。
表 2 ADC及SCC多因素Logistic回归分析结果Table 2. Results of Multifactor Logistic Regression Analysis of ADC and SCC变量 β SE Wald $\chi^2 $ OR 95%CI Z P CEA 0.005 0.003 1.880 1.005 (1.000-1.010) 1.987 0.047 分叶征 1.992 0.719 4.712 7.328 (1.931-33.680) 2.768 0.006 胸膜牵拉 1.413 0.599 2.883 4.109 (1.340-14.58) 2.359 0.018 毛刺征 1.466 0.736 2.346 4.333 (1.103-20.300) 1.993 0.046 常数 -5.977 1.355 0.587 0.003 (0.000-0.025) -4.410 <0.001 2.3 影像组学模型构建
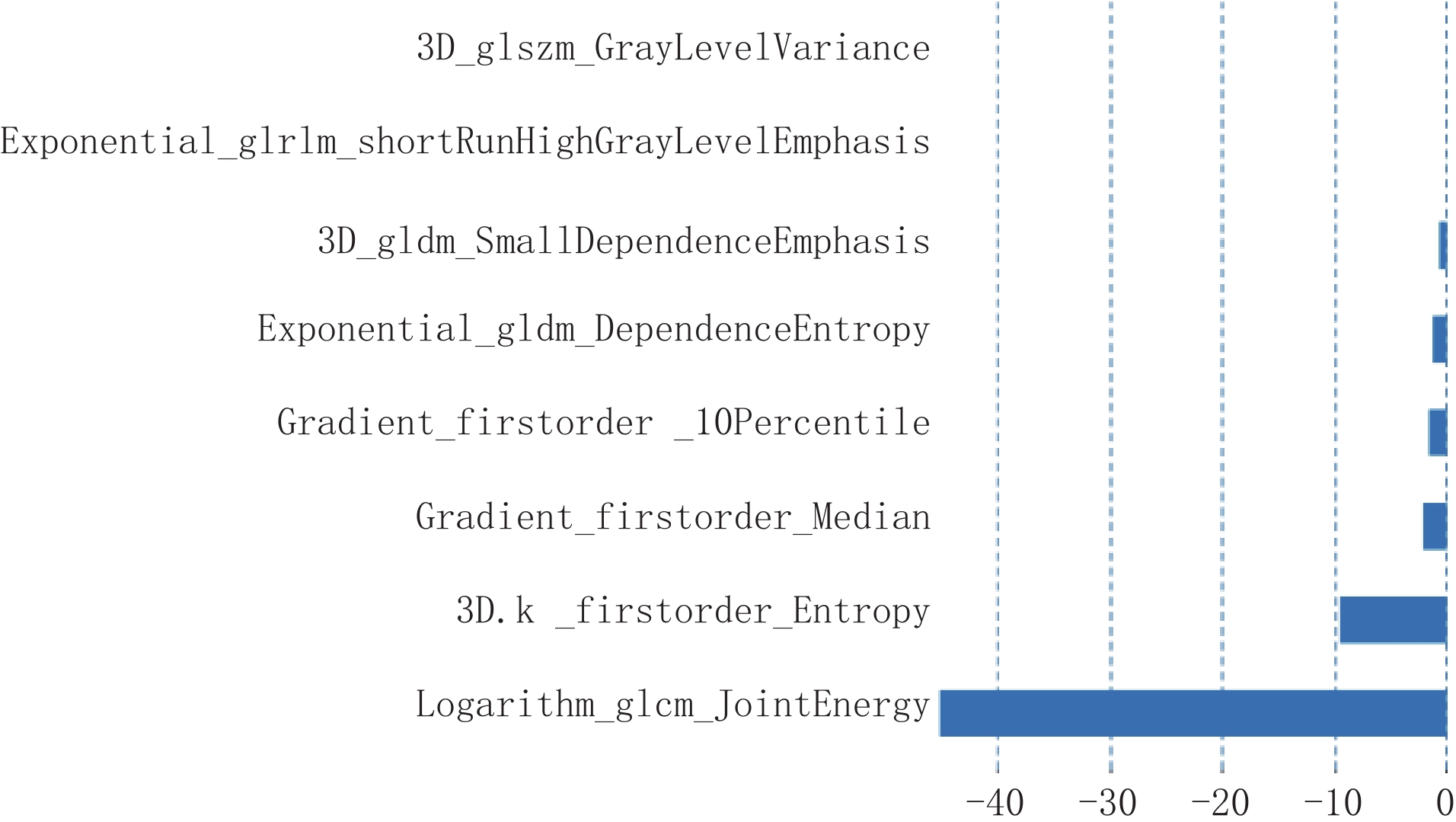
在训练集中,基于动脉期图像共提取了786个影像组学特征,经过ICC、Boruta及LASSO共筛选出来8个最优影像组学特征,分别为:一阶特征3个、二阶特征5个:灰度共生矩阵 (GLCM)特征1个、灰度区域大小矩阵(GLSZM)特征1个、灰度游程矩阵(GLRLM)特征1个和灰度依赖矩阵(GLDM)2个(图3)。将该8个影像组学特征构建影像组学标签,其影像组学评分为:
Rad-Score =2.362-1.613×3D_glszm_GrayLevelVariance+6.717×exponential_glrlm_ShortRunHighGrayLevelEmphasis+1.273×3D_gldm_SmallDependenceEmphasis-2.135×exponential_gldm_DependenceEntropy-1.621×gradient_firstorder_10Percentile+9.568×gradient_firstorder_Median-4.510×3D.k_firstorder_Entropy-3.288×logarithm_glcm_JointEnergy。
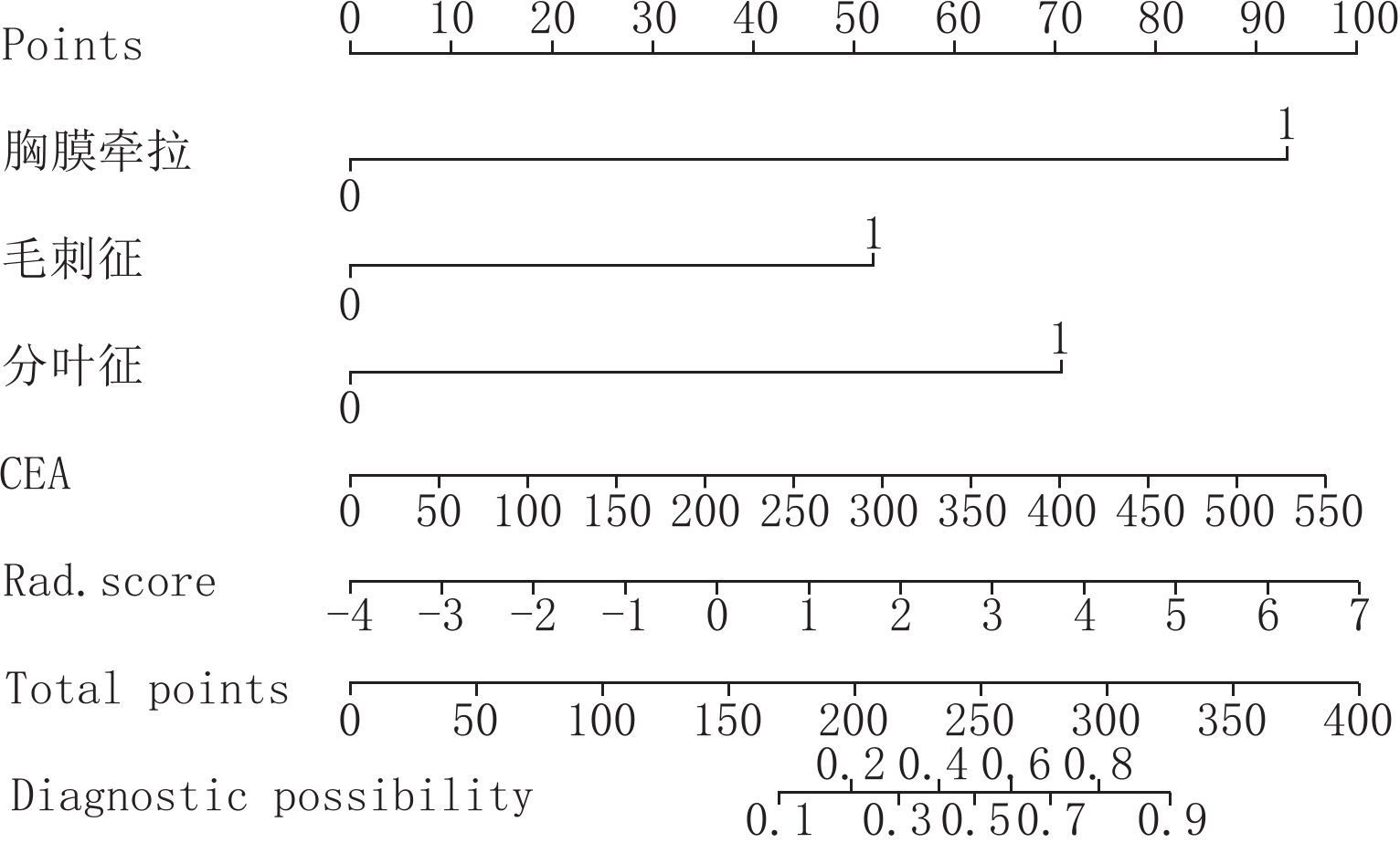
2.4 列线图模型构建
将CEA、分叶征、胸膜牵拉及毛刺征与Rad-Score 采用逻辑回归构建联合模型,并绘制列线图(图4)。
2.5 三种模型诊断效能评价与比较
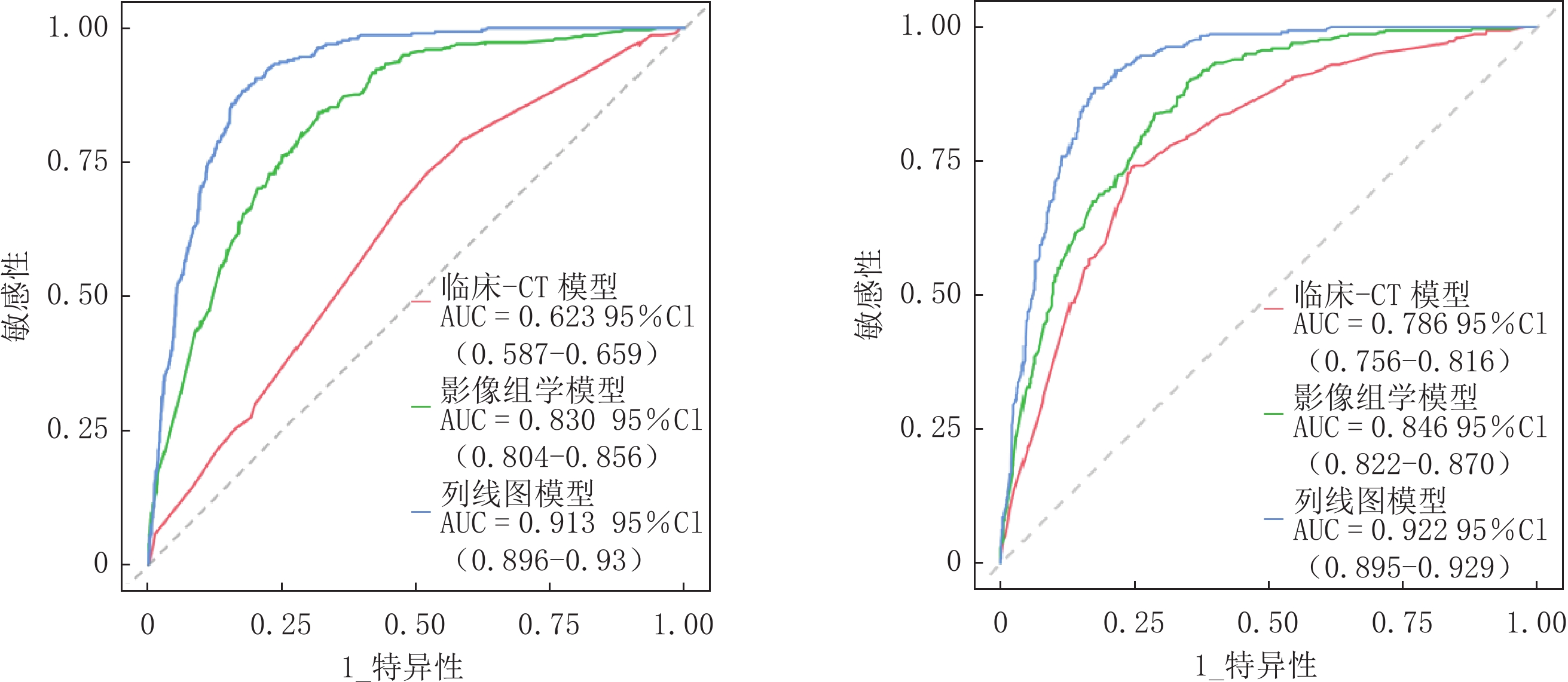
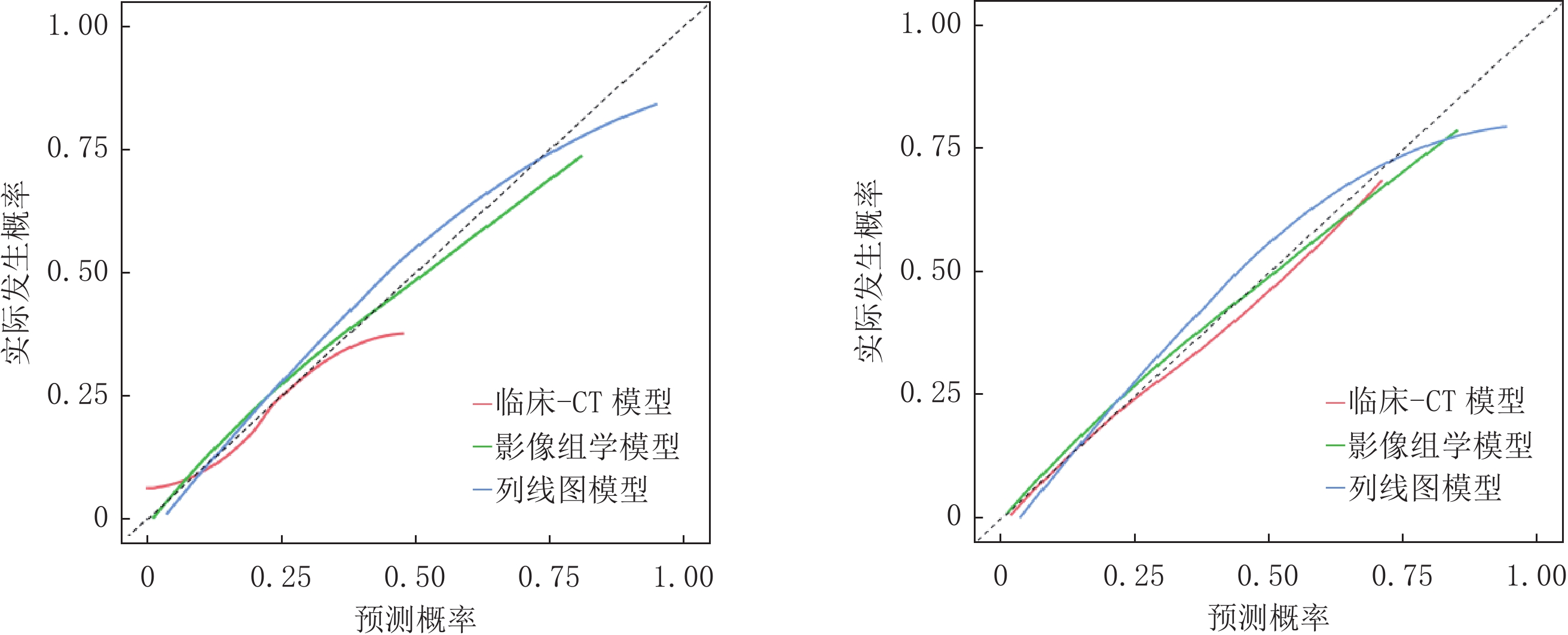
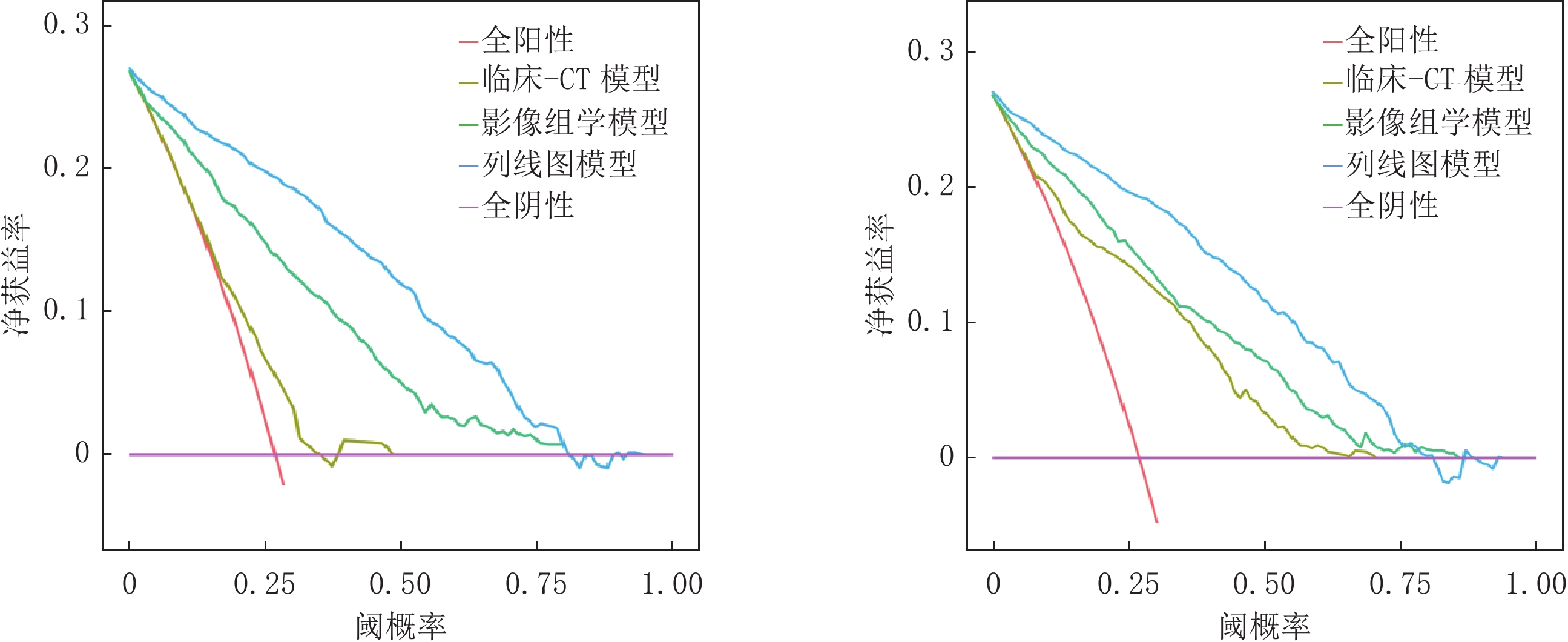
经ROC曲线分析显示,训练集及验证集中列线图模型AUC均最大,分别为0.913和0.922均高于影像组学模型(AUC=0.830、0.846)和临床-CT模型(AUC=0.623、0.786),差异均有统计学意义(训练集:Z= -2.213、-2.269,P=0.027、0.023;验证集:Z= -5.887 、-10.430 ,均P<0.001);影像组学模型验证集AUC高于临床-CT模型,差异有统计学意义(Z=3.016,P=0.038)(表3、图5)。Hosmer-Lemeshow检验结果显示,验证集临床-CT模型、影像组学模型及列线图模型的拟合度均良好(
$\chi^2 $ =3.173、12.437和8.528,P=0.923、0.133和0.476);校准曲线分析显示,训练集及验证集列线图模型的预测概率曲线与理想曲线最接近,优于其它两个模型(图6);DCA分析结果显示,训练集及验证集列线图模型的曲线下面积最大,临床净收益最高(图7)。表 3 临床-CT模型、影像组学模型及列线图模型诊断效能比较(%)Table 3. Comparison of diagnostic efficacy of clinical-CT model, radiomics model, and nomogram model(%)数据集 模型 AUC (95% CI) 敏感性 特异性 阳性预测值 阴性预测值 训练集 临床-CT模型 0.623(0.587~0.659) 76.79 71.82 76.58 75.45 影像组学模型 0.830(0.804~0.856) 83.78 80.63 81.55 79.92 列线图模型 0.913(0.896~0.930) 92.59 88.74 90.77 86.01 验证集 临床-CT模型 0.786(0.756~0.816) 73.83 75.58 73.53 88.34 影像组学模型 0.846(0.822~0.870) 83.89 81.10 82.52 92.05 列线图模型 0.922(0.895~0.929) 93.01 82.48 89.84 90.99 3. 讨论
肺癌的发病率在我国乃至全球一直处于高发状态,据相关研究表明,在每年新确诊的肺癌中非小细胞肺癌约占80%~85%,其中主要以鳞癌和腺癌为主,约占90%[9]。CT已被临床普遍用于肺癌的诊断,但是单独通过CT的影像特征很难对肺癌的病理类型做出准确有效的诊断。肺癌在不断生长发展的过程中会不断产生一种或几种正常状态下组织不含有的特异性物质,这些物质会反应出肿瘤的某些特征,临床中常通过这些肿瘤血清指标对肺癌病理类型进行辅助鉴别诊断[10-11]。影像组学是利用计算机深度挖掘技术对病灶进行高通量提取及筛选影像特征,能够无创定量反应出肿瘤内病理生理信息,以达到鉴别肿瘤类型的目的[12]。
肺癌的CT影像学特征主要表现为肿瘤大小、有无毛刺征、有无分叶征、有无坏死空洞或空泡征等,不同病理类型的肺癌其影像特征也不尽相同[13]。王守玉[14]等研究表明,鳞癌分叶征明显多于腺癌,这可能是由于鳞癌是以膨胀性生长为主且生长较快,而腺癌主要以伏壁式生长为主,故分叶征主要多见于鳞癌。彭弘[15]等在对肺癌CT征象与其病理对照研究中发现,胸膜牵拉及毛刺征多见于腺癌,这主要与腺癌中的纤维组织及结缔组织成分较多有关。本研究结果表明,SCC组分叶征及坏死空洞征均多于ADC组(均P<0.05),而血管集束征、胸膜牵拉及毛刺征少于ADC组(P<0.05),经进一步分析发现,分叶征、胸膜牵拉及毛刺征是鉴别ADC与SCC的为独立影响因素,与张英惠[16]研究结果相符。
CEA、CA199及NSE等肿瘤标记物在肺癌患者血清中明显上升,其作为肺癌肿瘤标记物已被临床广泛应用。谭淦纹[17]等在肺癌影像征象与CA199及NSE表达的相关性研究结果表明,肺癌有无坏死空洞、毛刺征、深分叶征与CA199及NSE的表达有正相关性。陈宝华[18]等将血清CEA联合影像特征在肺癌临床分期及预后研究发现,CEA联合影像特征能够有效预测肺癌临床分期及预后情况。本研究结果表明,ADC组CEA高于SCC组(P<0.05),且是鉴别ADC与SCC的为独立危险因素,说明肺癌患者血清CEA水平可作为鉴别ADC与SCC的影像学辅助检查手段。另外,本研究将CEA与具有显著差异的肺癌影像特征采用逻辑回归构建临床-CT模型,该模型训练集及验证集的AUC值分别为0.623和0.786,敏感性、特异性分别为76.79%、71.82%和73.83%、75.58%,与肖蓉[19]等研究结果一致。
Zhu[20]等在129名肺癌患者中从人工勾画的肿瘤区域中提取了约 485个影像组学特征,最终选择了五个组学特征用于构建区分ADC和SCC的影像组学模型,训练集AUC为0.905,敏感性和特异性分别为83.00%、92.90%,验证集AUC为 0.893,敏感性和特异性分别为 82.80%、90.00%,说明影像组学在鉴别肺癌病理类型方面具有较高的诊断效能。唐聪聪[21]等采用影像组学对非小细胞肺癌病理分级的研究结果中显示,共提取1 878个影像组学特征,应用支持向量机对特征的重要性进行评估并进行降维筛选得到 20个组学特征,并建立组学模型,该模型训练集 AUC为0.851,准确率为80.35%;测试集AUC为0.833,准确率为90.90%。本研究基于胸部增强动脉期图像共提取786个影像组学特征,包括一阶特征3个,二阶特征5个,依次采用ICC、Boruta及LASSO得到最优8个影像组学特征,并建立影像组学模型,结果表明该影像组学模型训练集和验证集的AUC分别为0.830和0.846。最后,本研究将独立影响SCC与ADC的临床-CT因素与影像组学标签Rad-score构建联合列线图模型,其训练集和验证集AUC分别为0.913和0.922,均高于影像组学模型和临床-CT模型(均P <0.05);方家杨等[22]基于临床、影像组学和计算机视觉特征构建的联合模型在鉴别SCC和ADC的研究结果表明,该联合模型在训练集和验证集的AUC分别为为 0.865 和 0.851,均高于单一模型(P<0.05);陆亮等[23]将影像组学特征联合CT形态学特征构建联合模型对SCC和ADC进行鉴别,结果表明,联合模型在训练集及验证集的AUC分别为 0.879、0.869,均高于单一CT临床模型。以上研究结果均与本研究结果均相符,说明联合影像组学特征、临床特征和CT特征构建的联合模型比单一模型在鉴别诊断肺癌病理分型中具有更佳的预测能力。另外,本研究经Hosmer-Lemeshow检验显示,3组模型拟合度均良好(均P>0.05);校准曲线及DCA分析显示,列线图模型的预测概率曲线及曲线下面积均优于其它两个模型,说明列线图联合模型对SCC与ADC的鉴别诊断能力最高,与汪靖婷[24]等研究结果相符。另外,本研究中根据联合模型绘制了列线图,其已广泛用于对肿瘤分型、诊断、淋巴结转移及预后效果评估,以可视化图形方式更加直观地反映每个预测因子对疾病未来发展结果的影响,对每位患者进行个性化分析及诊断[25-26]。
本研究仍存在一定的不足之处,(1)为单中心的回顾性研究,且样本量较小,数据结果可能存在一定偏倚;(2)仅提取了动脉期图像组学特征,未对平扫及静脉期图像组学特征进行研究;(3)仅从病灶最大层面提取组学特征,不能反映肿瘤的全部特征。
综上所述,临床-CT模型、影像组学模型及联合列线图模型对SCC与ADC均有一定的鉴别诊断能力,尤以联合列线图模型最佳,有望成为无创鉴别SCC与ADC的一种新的影像检查方法。
-
表 1 训练集两组患者临床资料与CT影像特征结果比较(
$\bar x\pm s $ )[n(%)]Table 1 Comparison of clinical data and results of CT imaging characteristics between the two groups of patients in the training set
指标 SCC组(n=29) ADC组(n=56) t/x2/Z P 年龄 72.21±9.27 67.84±9.64 1.206 0.144 性别 1.231 0.121 男 24(82.76) 28(50.00) 女 5(17.24) 28(50.00) 吸烟史 0.188 0.665 是 19(65.52) 34(60.71) 否 10(34.48) 22(39.29) 家族肿瘤史 0.197 0.657 是 12(41.38) 26(46.43) 否 17(58.62) 30(53.57) NSE(ng/mL) 4.56(3.04, 7.38) 6.24 (4.12, 8.33) 0.413 0.115 CEA(ng/ml) 96.53±8.15 198.22±21.51 6.495 <0.001 CA242(IU/ml) 28.42±4.37 37.49±5.84 1.037 0.189 CA199(U/mL) 51.17±9.53 65.27±10.51 0.202 0.565 坏死空洞 有 25(86.21) 13(23.21) 35.325 <0.001 无 2(6.89) 43(76.79) 毛刺征 有 10(34.48) 45(80.36) 17.606 <0.001 无 19(65.52) 11(19.64) 分叶征 有 26(89.66) 10(17.86) 40.340 <0.001 无 3(10.34) 46(82.14) 空泡征 有 22(75.86) 32(57.14) 2.890 0.089 无 7(24.14) 24(42.86) 胸膜牵拉 有 16(55.17) 44(78.57) 4.574 0.032 无 13(44.83) 12(21.43) 血管集束征 有 13(44.83) 41(73.21) 6.645 0.010 无 16(55.17) 15(26.79) 肿瘤位置 上叶 18(62.07) 30(53.57) 2.011 0.093 中叶 3(10.34) 9(16.07) 下叶 8(27.59) 17(30.36) 表 2 ADC及SCC多因素Logistic回归分析结果
Table 2 Results of Multifactor Logistic Regression Analysis of ADC and SCC
变量 β SE Wald $\chi^2 $ OR 95%CI Z P CEA 0.005 0.003 1.880 1.005 (1.000-1.010) 1.987 0.047 分叶征 1.992 0.719 4.712 7.328 (1.931-33.680) 2.768 0.006 胸膜牵拉 1.413 0.599 2.883 4.109 (1.340-14.58) 2.359 0.018 毛刺征 1.466 0.736 2.346 4.333 (1.103-20.300) 1.993 0.046 常数 -5.977 1.355 0.587 0.003 (0.000-0.025) -4.410 <0.001 表 3 临床-CT模型、影像组学模型及列线图模型诊断效能比较(%)
Table 3 Comparison of diagnostic efficacy of clinical-CT model, radiomics model, and nomogram model(%)
数据集 模型 AUC (95% CI) 敏感性 特异性 阳性预测值 阴性预测值 训练集 临床-CT模型 0.623(0.587~0.659) 76.79 71.82 76.58 75.45 影像组学模型 0.830(0.804~0.856) 83.78 80.63 81.55 79.92 列线图模型 0.913(0.896~0.930) 92.59 88.74 90.77 86.01 验证集 临床-CT模型 0.786(0.756~0.816) 73.83 75.58 73.53 88.34 影像组学模型 0.846(0.822~0.870) 83.89 81.10 82.52 92.05 列线图模型 0.922(0.895~0.929) 93.01 82.48 89.84 90.99 -
[1] 傅睿, 吴一龙, 钟文昭. 2020版肺癌多学科团队诊疗中国专家共识解读[J]. 中国肿瘤临床, 2022, 49(4): 163−167. DOI: 10.12354/j.issn.1000-8179.2022.20211201. FU R, WU Y L, ZHONG W Z. Interpretation of Chinese expert consensus on multidisciplinary team diagnosis and treatment of lung cancer(2020 version)[J]. Chinese tumor clinical, 2022, 49(4): 163−167. DOI: 10.12354/j.issn.1000-8179.2022.20211201.
[2] 王馨慧, 张笑雪, 聂奔, 等. 联合免疫模式在晚期非小细胞肺癌治疗中的研究进展[J]. 临床肿瘤学杂志, 2023, 28(3): 263−269. DOI: 10.3969/j.issn.1009-0460.2023.03.011. WANG X H, ZHANG X X, NIE B, et al. Research progress of combined immunotherapy mode in the treatment of advanced non-small cell lung cancer[J]. Chinese Clinical Oncology, 2023, 28(3): 263−269. DOI: 10.3969/j.issn.1009-0460.2023.03.011.
[3] 吕祥瑞, 皇甫娟, 王孟丽, 等. 血清肿瘤标志物与肺癌病理类型的相关性研究[J]. 癌症进展, 2021, 19(14): 1451−1455. DOI: 10.11877/j.issn.1672-1535.2021.19.14.13. LV X R, HUANG P J, WANG M L, et al. Study on the correlation between serum tumor markers and pathological types of lung cancer[J]. Oncology Progress, 2021, 19(14): 1451−1455. DOI: 10.11877/j.issn.1672-1535.2021.19.14.13.
[4] 张盼, 黄庆, 汪扬, 等. 非小细胞肺癌动态增强CT扫描下临床表现特征与其病理类型的关系[J]. 西部医学, 2023, 35(4): 584-587. DOI: 10.3969/j.issn.1672-3511.2023.04.023. ZHANG P, HUANG Q, WANG Y, et al. Clinical characteristics of dynamic contrast-enhanced CT scan and their relationship with pathological types in non-small cell lung cancer[J]. Western medicine, [4]2023, 35(4): 584-587. DOI:10.3969/j.issn.1672-3511.2023.04.023.(in Chinese).
[5] 陈杰, 洪悦, 王艳, 等. 基于CT平扫影像组学特征在预测胸腺上皮性肿瘤WHO简化病理分型中的价值[J]. 中国CT和MRI杂志, 2024, 22(1): 71−73. DOI: 10.3969/j.issn.1672-5131.2024.01.023. CHEN J, HONG Y, WANG Y, et al. Value of Simplified Pathological Classification of Thymus Epithelial Tumors Based on Plain CT Imaging Features[J]. Chinese Journal of CT and MRI, 2024, 22(1): 71−73. DOI: 10.3969/j.issn.1672-5131.2024.01.023.
[6] 党俊明, 朱超华, 黄慧娴, 等. 基于MRI影像组学特征预测宫颈腺癌与鳞癌病理分型的研究[J]. 医疗卫生装备, 2022, 43(5): 54−59. DOI: 10.19745/j.1003-8868.2022099. DANG J M, ZHU C H, HUANG X H, et al. Prediction of pathological classification of cervical adenocarcinoma and squamous cell carcinoma based on MRI radiomics characteristics[J]. Medical and Health equipment, 2022, 43(5): 54−59. DOI: 10.19745/j.1003-8868.2022099.
[7] 高琳, 于鑫鑫, 康冰, 等. CT 影像组学对肺纯磨玻璃结节浸润性的预测价值[J]. 山东大学学报 (医学版), 2022, 60(5): 87−97. DOI: 10.6040/j.issn.1671-7554.0.2022.0500. GAO L, YU X X, KANG B, et al. Predictive value of CT-based radiomics nomogram for the invasiveness of lung pure ground-glass nodules[J]. Journal of ShanDong University(Health Sciences), 2022, 60(5): 87−97. DOI: 10.6040/j.issn.1671-7554.0.2022.0500.
[8] 中华医学会, 中华医学会肿瘤学分会, 中华医学会杂志社. 中华医学会肺癌临床诊疗指南(2019版)[J]. 中华肿瘤杂志, 2020, 42(4): 257−287. DOI: 10.3760/cma.j.cn112152-20200120-00049. Chinese Medical Association, Chinese Medical Association Oncology Branch, Journal of Chinese Medical Association. Chinese Medical Association guidelines for clinical diagnosis and treatment of lung cancer (2019 edition)[J]. Chinese Journal of Tumors, 2020, 42(4): 257−287. DOI: 10.3760/cma.j.cn112152-20200120-00049.
[9] 朱玲玲, 伍娟, 王艇, 等. 2023年第2版NCCN肺癌筛查临床实践指南更新解读[J]. 实用肿瘤杂志, 2023, 38(5): 399−407. DOI: 10.13267/j.cnki.syzlzz.2023.063. ZHU L L, WU J, WANG T, et al. Updated interpretation of NCCN clinical practice guidelines for lung cancer screening, version 2.2023[J]. Journal of Practical Oncology, 2023, 38(5): 399−407. DOI: 10.13267/j.cnki.syzlzz.2023.063.
[10] 蒋会东, 谢强, 李军, 等. 64排CT联合NSE、ProGRP在肺癌鉴别诊断及TNM分期中的应用[J]. 中国现代医学杂志, 2022, 32(14): 95−100. DOI: 10.3969/j.issn.1005-8982.2022.14.017. JIANG H D, XIE Q, LI J, et al. Application of 64-detector-row CT combined with NSE and ProGRP in differential diagnosis and TNM staging of lung cancer[J]. China Journal of Modern Medicine, 2022, 32(14): 95−100. DOI: 10.3969/j.issn.1005-8982.2022.14.017.
[11] 孔令芹, 陈苏媛, 李西川, 等. 早中期小细胞肺癌与非小细胞肺癌CT影像及肿瘤标记物对比[J]. 中国医学创新, 2023, 20(17): 42−46. DOI: 10.3969/j.issn.1674-4985.2023.17.010. KONG L Q, CHEN S Y, LI X C, et al. Comparison of CT Images and Tumor Markers between Early and Middle Stage Small Cell Lung Cancer and Non-small Cell Lung Cancer[J]. MedicaI Innovation of China, 2023, 20(17): 42−46. DOI: 10.3969/j.issn.1674-4985.2023.17.010.
[12] 唐新, 梁江涛, 向柏林, 等. PET/MRI影像组学特征预测肺腺癌与肺鳞癌病理分型价值[J]. 浙江医学, 2022, 44(6): 580−584. DOI: 10.12056/j.issn.1006-2785.2022.44.6.2021-2731. TANG X, LIANG J T, XIANG B L, et al. Application of PET/MRI radiomics in differentiating the pathological types of lung adenocarcinoma and squamous cell carcinoma[J]. Zhejiang medicine, 2022, 44(6): 580−584. DOI: 10.12056/j.issn.1006-2785.2022.44.6.2021-2731.
[13] 孙士鹤, 侯艳娟, 刘芬, 等. CT诊断老年肺癌患者病理亚型与病理学诊断的一致性及其影像学特征[J]. 中国老年学杂志, 2023, 43(24): 5915−5918. DOI: 10.3969/j.issn.1005-9202.2023.24.007. SUN S H, HOU Y J, LIU F, et al. Concordance between pathologic subtypes and pathologic diagnoses and their imaging features in CT-diagnosed elderly patients with lung cancer[J]. Chinese Journal of Gerontology, 2023, 43(24): 5915−5918. DOI: 10.3969/j.issn.1005-9202.2023.24.007.
[14] 王守玉, 屈开新, 王庆龙, 等. 不典型肺癌CT诊断与鉴别诊断[J]. 中国医药科学, 2020, 10(15): 187−190. DOI: 10.3969/j.issn.2095-0616.2020.15.051. WANG S Y, QU K X, WANG Q L, et al. CT diagnosis and differential diagnosis of atypical lung cancer[J]. China Medicine and Pharmacy, 2020, 10(15): 187−190. DOI: 10.3969/j.issn.2095-0616.2020.15.051.
[15] 彭弘, 李圣博, 赵旭. 周围型肺癌CT征象与病理的对照相关性研究[J]. 罕少疾病杂志, 2022, 29(12): 42−43. DOI: 10.3969/j.issn.1009-3257.2022.12.019. PENG H, LI S B, ZHAO X. A Comparative Study of CT Signs and Pathology of Peripheral Lung Cancer[J]. Journal of Rare and Uncommon Diseases, 2022, 29(12): 42−43. DOI: 10.3969/j.issn.1009-3257.2022.12.019.
[16] 张英惠. 96例早期周围型肺癌的CT影像特征分析[J]. 中国现代药物应用, 2022, 16(4): 87-90. DOI: 10.14164/j.cnki.cn11-5581/r.2022.04.033. ZHANG Y H. Analysis of CT imaging features of 96 cases of early-stage peripheral lung cancer[J]. Chinese Modern Drug Application, [16]2022, 16(4): 87-90. DOI:10.14164/j.cnki.cn11-5581/r.2022.04.033.(in Chinese).
[17] 谭淦纹, 曹欢, 王玲. 肺癌MSCT增强扫描影像学征象与CA199、CA242、NSE表达的相关性及临床应用价值研究[J]. 中国CT和MRI杂志, 2022, 20(3): 49−51. DOI: 10.3969/j.issn.1672-5131.2022.03.016. TAN J W, CAO H, WANG L. Correlation between Imaging Signs of MSCT Enhanced Scanning of Lung Cancer and the Expression of CA199, CA242, NSE, and Its Clinical Application Value[J]. Chi nese Journal of CT and MRI, 2022, 20(3): 49−51. DOI: 10.3969/j.issn.1672-5131.2022.03.016.
[18] 陈宝华, 段强军. 血清 CEA、NSE 联合影像学特征在肺癌分期及预后中的价值[J]. 分子诊断与治疗杂志, 2022, 14(9): 1615−1619. DOI: 10.3969/j.issn.1674-6929.2022.09.040. CHEN B H, DUANG Q J. The value of serum CEA and NSE combined with imaging features in lung cancer staging and prognosis[J]. Journal of Molecular Diagnosis and Treatment, 2022, 14(9): 1615−1619. DOI:10.3969/j.i ssn. 1674-6929.2022.09.040. DOI: 10.3969/j.issn.1674-6929.2022.09.040. (in Chinese).
[19] 肖蓉, 潘频华. 老年肺癌CT影像学特征与特异性标记物的相关性研究及联合诊断[J]. 国际老年医学杂志, 2020, 41(6): 391−394. DOI: 10.3969/j.issn.1674-7593.2020.06.013. XIAO R, PAN P H. Correlation between CT Imaging Features and Specific Markers and Their Combined Diagnostic Value in Lung Cancer in Older Patients[J]. International Journal of Geriatrics, 2020, 41(6): 391−394. DOI: 10.3969/j.issn.1674-7593.2020.06.013.
[20] Zhu X, Dong D, Chen Z, et al. Radiomic signature as a diagnostic factor for histologic subtype classification of non-small cell lung cancer[J]. Eur Radiol, 28(7): 2772-2778. DOI: 10.1007/s00330-017-5221-1.
[21] 唐聪聪, 陈艾琪, 曹胜男, 等. CT 影像组学在非小细胞肺癌病理分级中的应用[J]. 蚌埠医学院学报, 2023, 48(6): 783-786. DOI: 10.13898/j.cnki.issn.1000-2200.2023.06.017. TANG C C, CHEN A Q, CAO S N, et al. Application of CT radiomics in the pathological grading of non-small cell lung cancer[J]. Journal of Bengbu Medical College, [21]2023, 48(6): 783-786. DOI:10.13898/j.cnki.issn.1000-2200.2023.06.017.(in Chinese).
[22] 方家杨, 梁长宇, 陶俊利, 等. 基于临床、影像组学和计算机视觉特征鉴别肺鳞癌和腺癌[J]. 中国医学影像学杂志, 2022, 30(7): 691−696,702. DOI: 10.3969/j.issn.1005-5185.2022.07.010. FANG J Y, LIANG C Y, TAO J L, et al. Differentiation of Lung Squamous Cell Carcinoma and Adenocarcinoma Based on Clinical, Radiomic and Computer Vision Features[J]. Chinese Journal of Medical Imaging, 2022, 30(7): 691−696,702. DOI: 10.3969/j.issn.1005-5185.2022.07.010.
[23] 陆亮, 徐圆. 影像组学联合CT特征对周围型肺腺癌及鳞癌的鉴别价值研究[J]. 医疗卫生装备, 2021, 42(10): 48−52,63. DOI: 10.19745/j.1003-8868.2021211. LU L, XU Y. Diagnostic value of radiomics combined with CT features for differentia-ting peripheral lung adenocarcinoma from squamous cell carcinoma[J]. Chinese Medical Equipment Journal, 2021, 42(10): 48−52,63. DOI: 10.19745/j.1003-8868.2021211.
[24] 汪靖婷, 钟飞扬, 甘甜, 等. 基于临床及 CT 影像组学特征构建周围型小细胞肺癌与肺腺癌诊断模型的研究[J]. 临床放射学杂志, 2023, 42(3): 406-410. DOI: 10.13437/j.cnki.jcr.2023.03.035. WANG T T, ZHONG F Y, GAN T, et al. Study on the Differential Diagnosis Model between Periph eral SCLC and ADC Based on Clinical and CT Radiomics Features[J]Journal of Clinical Radio logy, 2023, 42(3): 406-410. DOI:10.13437/j.cnki.jcr.2023.03.035.(in Chinese).
[25] Wen C L, Meng P L, Wen Y H, et al. A practical dynamic nomogram model for predicting bone metastasis in patients with thyroid cancer[J]. Front Endocrinol (Lausanne), 2023, 14: 1142796. DOI: 10.3389/fendo.2023.1142796.
[26] Feng H, Yu Q S, Wang J X, et al. Establishment and validation of nomogram prediction model for complicated acute appendicitis[J]. Zhonghua Wai Ke Za Zhi, 2023, 61(12): 1074−1079. DOI: 10.3760/cma.j.cn112139-20230104-00005.



 下载:
下载: